Is the FDA about to approve an experimental COVID vaccine for kids?
Private profits over public health.
On Tuesday, October 26, 2021, the FDA is meeting “to discuss a request to amend Pfizer-BioNTech’s Emergency Use Authorization (EUA) for administration of their COVID-19 mRNA vaccine to children 5 through 11 years of age.”
In anticipation of this meeting (and to illuminate the real reason why Pfizer is seeking the EUA), we offer a rare point of agreement in our polarized world. One that damns both the FDA and Pfizer: healthy children aren’t threatened by COVID-19.
As compared to adults, children are less prone to get COVID, they are less prone to be hospitalized by COVID, and they are far less likely to die from COVID.
This is supported by CDC data (quoted by Pfizer), which shows that for children aged 5-11 years-old, there have been 1.8 million COVID-19 cases and only 138 deaths. By comparison, approximately double that amount of children have died of pneumonia during this same time period. For those children hospitalized with COVID-19, the vast majority had more than one underlying condition (chronic lung disease, obesity, or neurologic disorders).
According to NY Magazine, “among children the mortality risk from COVID-19 is actually lower than from the flu. The risk of severe disease or hospitalization is about the same.”
One study published in Nature estimated “that the infection fatality ration [of COVID-19] is lowest among 5–9-year-old children.” That same publication has noted that children just aren’t safe from serious COVID-19 infection. Children “aren’t driving viral spread” and they don’t get infected or spread the infection like adults. For example, studies of some schools in Nebraska showed “were open the whole year with over 20,000 students and staff, and there were only 2 transmission events during that entire study period.”
The CDC concedes that “children are less likely to develop severe illness or die from COVID-19.” It notes that “findings from several studies suggest that SARS-CoV-2 transmission among students is relatively rare, particularly when prevention strategies are in place.” It reluctantly admits “several contact tracing studies have found limited student-to-student transmission in schools.”
These facts would seem to contradict the the legal and factual justifications necessary for giving the vaccine an EUA for kids aged 5-11. For review, the FDA’s issuance of an EUA is governed by 21 U.S.C. 360bbb-3. In its original issuance of the EUA for the Pfizer vaccine back in 2020, the FDA concluded:
One can’t help but observe, based on the extremely low numbers of child deaths, that there are “adequate, approved, and available alternative[s]” to the Pfizer vaccine. Even for those children hospitalized with COVID-19, their recovery is sufficient proof that there are effective treatments out there. The data is clear that there is no justification for the FDA to issue this particular EUA.
And if COVID-19 constituted a serious public health emergency for children, why didn’t Pfizer focus on this age group last year?
Anyway, we make these observations and discuss the facts but we’re not sure how much it matters. It seems the FDA has already reached its decision on the necessity for a vaccine for children 5-11 years of age. The October 26 meeting would just be a pretext, the last hoop it has to jump through before vaccines are mandated in schools around the country. As the FDA has already stated, “the benefits of the Pfizer-BioNTech COVID-19 Vaccine 2-dose primary series clearly outweigh the risks for ages 5-11 years.”
This FDA statement is made as the authorities promise kids will get the vaccine. On Sunday, Dr. Anthony Fauci guessed the vaccine would be available by “early November.”

This came just over two weeks after the prediction from Pfizer’s Scott Gottlieb, M.D., who said the FDA go through a quick review of “weeks not months.” And Dr. Fauci’s comments come as the Biden White House promises to help vaccinate all eligible children. According to House Vaccination Coordinator Dr. Bechara Choucair:
“We know we have about 28 million kids five to eleven in this country, and we have to be ready. We’ve been spending a lot of time making sure that we have tens of thousands of family [doctors] and pediatricians ready to administer vaccines at their offices.”
The Pfizer Data
Pfizer has released its data for the clinical studies of its vaccines being administered to kids aged 5-11. Here’s what you need to know.
Pfizer boasts a “vaccine efficacy of 90.7%” – in the short-term. It did not measure the vaccine’s long-term effectiveness.
The number of participants in the Pfizer studies was (conveniently) “too small to detect any potential risks of myocarditis associated with vaccination.”
The long-term safety of the Pfizer COVID-19 vaccine in kids aged 5-11 is unknown and will be studied at a later time. (According to Pfizer, “Long-term safety of COVID-19 vaccine in participants 5 to <12 years of age will be studied in 5 post-authorization safety studies.”)
Pfizer concludes the benefits of its vaccine “outweigh the known or potential risks” – while admitting it does not know the long-term risks.
Pfizer previously announced the vaccine’s side effects for kids aged 5-11 is “generally comparable to those observed in participants 16 to 25 years of age.” This is concerning because young men within that age-range have the highest incident of myocarditis after the second Pfizer dose.
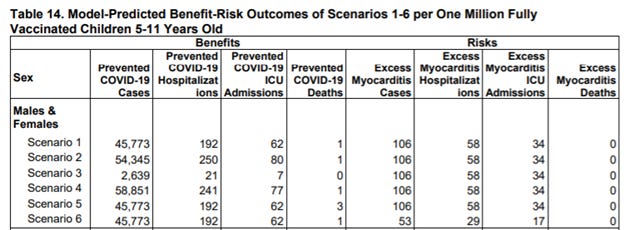
Despite the unknowns, the FDA has concluded the benefits of the vaccine “clearly outweigh” the risks. But is that really the case? Look for yourself. Here are the FDA calculations, with most its models predicting only ONE preventable COVID-19 death per million children vaccinated. While it estimates COVID-19 ICU admissions will go down, those numbers aren’t much better than the excess myocarditis ICU admissions to be caused by the Pfizer vaccine.
The Upcoming Vote on the Pfizer Vaccine
There are also concerns with the FDA committee members. Dr. James Hildreth (a temporary voting member) is a professor and president/CEO of Meharry Medical College in Tennessee. He submitted this “Acknowledgement of Financial Interests”:
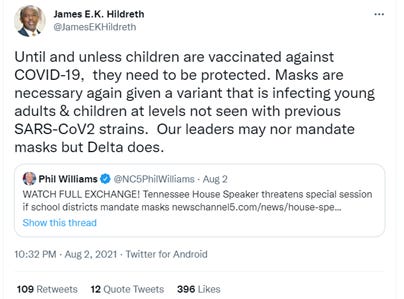
While those are mostly institutional interests (“imputed interests”) that benefit Dr. Hildreth, perhaps more concerning is the fact that he’s already on the record supporting the need to vaccinate children.
And as to the broader issue of personal autonomy – the concept that people should not be subjected to experimental treatment – here’s Dr. Hildreth advocating “mandatory vaccines.”
Pfizer Motivations
Finally, if all the experts are correct – if there is no COVID-19 emergency for kids 5-11 years old – then why is Pfizer seeking an emergency use authorization?
It seems the answer lies in the profits and not the public health.
And if that’s the case, then the FDA is corrupted for good.





Parents will fight this to the very end. This is the hill.
The children will be the tipping point if this is mandated. I get a lot of ppl telling me what’s the difference if they mandated for measles mumps ect . They difference is the years of testing that we don’t have yet. Will definitely be pulling kids out of school